A CTMS (Clinical Trial Management System) is software used by pharma companies, CROs, and research organizations to manage clinical trials efficiently. It centralizes planning, tracking, compliance, and reporting. This guide explains features, benefits, use cases, pricing, and the best CTMS platforms across different countries.
Table of Contents
What is CTMS?
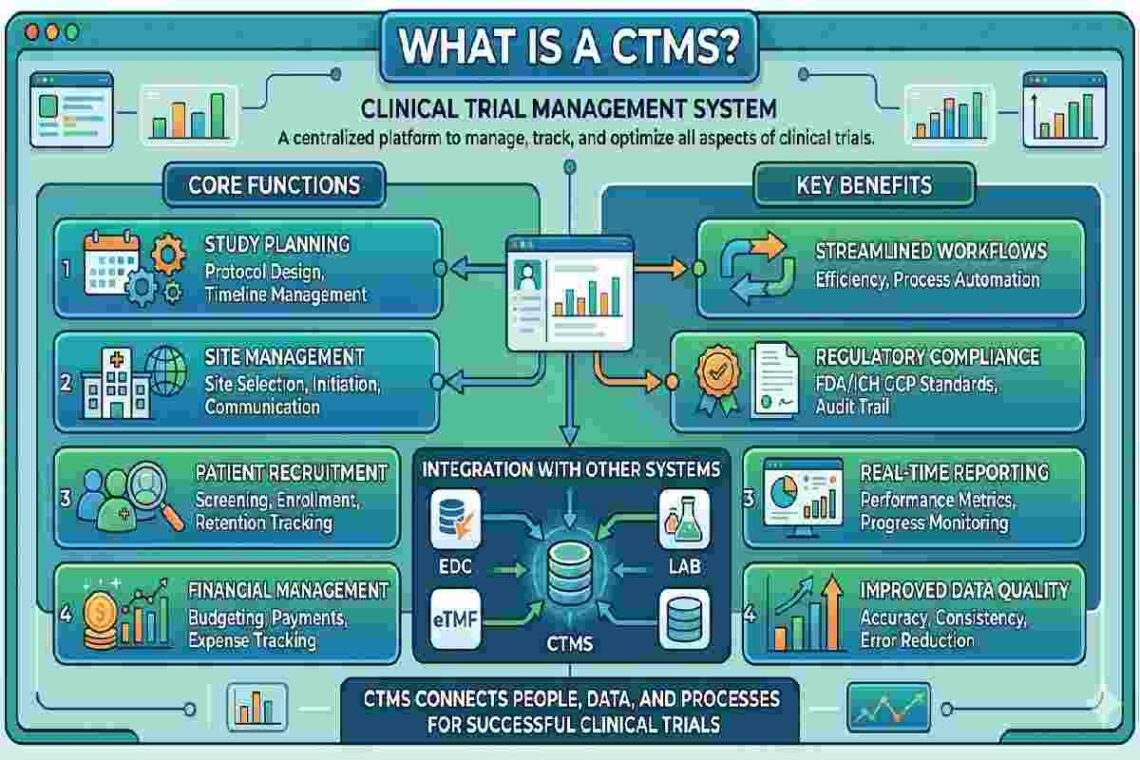
A Clinical Trial Management System (CTMS) is a specialized software platform designed to plan, manage, track, and optimize clinical trials from start to finish.
It helps organizations:
- Manage study timelines and milestones
- Track patient recruitment and enrollment
- Monitor budgets and payments
- Ensure regulatory compliance
- Generate reports and analytics
CTMS is widely used by:
- Pharmaceutical companies
- Biotechnology firms
- Contract Research Organizations (CROs)
- Hospitals and research institutions
Why CTMS Is Critical in Modern Clinical Trials
Clinical trials are becoming increasingly complex due to:
- Multi-site global studies
- Strict regulatory requirements
- Large datasets and patient tracking

Key Benefits
Centralized data management
Improved compliance and audit readiness
Faster trial execution
Better collaboration across teams
Real-time reporting and insights
Core Features of a CTMS
1. Study Planning & Management
- Protocol tracking
- Milestone scheduling
- Site selection
2. Patient Recruitment Tracking
- Enrollment status
- Screening logs
- Retention monitoring
3. Financial Management
- Budget tracking
- Payments to sites
- Invoice management
4. Regulatory Compliance
- Document management
- Audit trails
- Compliance tracking (FDA, EMA, etc.)
5. Reporting & Analytics
- Custom dashboards
- KPI tracking
- Performance insights
Types of CTMS Solutions
| Type of CTMS | Description | Key Features | Best For | Pros | Cons |
| Enterprise CTMS | Comprehensive, large-scale systems designed for global clinical trials | Advanced analytics, multi-site management, integrations (EDC, eTMF), compliance tools | Large pharmaceutical companies | Highly scalable, robust features, strong compliance | Expensive, complex implementation |
| Cloud-Based (SaaS) CTMS | Hosted online and accessible via web browsers | Remote access, automatic updates, subscription pricing, data backups | CROs, startups, mid-sized companies | Cost-effective, easy to deploy, scalable | Data security concerns (if misconfigured), limited customization |
| On-Premise CTMS | Installed and managed on company servers | Full data control, custom configurations, high security | Large enterprises with strict data policies | Maximum control, strong data privacy | High setup cost, maintenance-heavy |
| Site CTMS | Designed specifically for clinical trial sites (hospitals, clinics) | Patient tracking, scheduling, financial tracking, site workflows | Research sites, hospitals | User-friendly, tailored for site needs | Limited enterprise-level features |
| Open-Source CTMS | Free or low-cost customizable platforms | Basic trial management, customizable code, community support | Small organizations, academic research | Low cost, flexible customization | Limited support, requires technical expertise |
| Hybrid CTMS | Combines cloud and on-premise features | Flexible deployment, data sync, customizable modules | Organizations needing flexibility | Balanced control and accessibility | Complex setup and integration |
| Mobile CTMS | Optimized for mobile devices and remote trial management | Real-time updates, mobile dashboards, field data access | Decentralized trials, remote teams | High accessibility, supports DCT (Decentralized Clinical Trials) | Limited full-feature capabilities vs desktop |
| Integrated CTMS (All-in-One) | Bundled with EDC, eTMF, and other clinical tools | Unified platform, seamless data flow, centralized reporting | Pharma companies, CROs | Eliminates data silos, efficient workflows | Vendor lock-in, higher cost |
CTMS vs EDC vs eTMF
| Feature | CTMS (Clinical Trial Management System) | EDC (Electronic Data Capture) | eTMF (Electronic Trial Master File) |
| Primary Purpose | Manage and track clinical trial operations | Collect and manage clinical trial data | Store and manage regulatory documents |
| Core Function | Operational oversight and planning | Data collection from patients/sites | Document storage and compliance |
| Main Users | Sponsors, CROs, project managers | Investigators, site staff, data managers | Regulatory teams, QA teams, auditors |
| Data Type | Operational data (timelines, budgets, sites) | Clinical data (patient records, outcomes) | Documents (protocols, consent forms, reports) |
| Key Features | Study planning, site tracking, budgeting, reporting | eCRFs, data validation, query management | Document versioning, audit trails, compliance tracking |
| Focus Area | Trial management & execution | Data accuracy & integrity | Regulatory compliance & documentation |
| Workflow Role | Oversees entire trial lifecycle | Captures patient-level data during trial | Maintains documentation for audits |
| Regulatory Role | Ensures operational compliance | Ensures data integrity (FDA, GCP) | Ensures audit readiness and document compliance |
| Integration | Integrates with EDC, eTMF, and other systems | Integrates with CTMS and eTMF | Integrates with CTMS and EDC |
| Example Tasks | Track enrollment progress, manage budgets | Record patient visits, lab results | Store consent forms, protocols, approvals |
| Real-Time Updates | Yes (operational dashboards) | Yes (live data entry and validation) | Yes (document updates and tracking) |
| End Users Goal | Efficient trial execution | Accurate data collection | Complete and compliant documentation |
| Complexity Level | Medium to High | High (data-intensive) | Medium |
| Best For | Managing trial logistics | Handling clinical data | Managing regulatory documents |
How to Choose the Best CTMS
Key Factors to Consider
- Scalability
- Can it handle multi-country trials?
- Integration
- Works with EDC, eTMF, and CRM systems?
- Compliance
- Meets FDA, HIPAA, GDPR standards?
- Ease of Use
- User-friendly interface for teams
- Cost
- Subscription vs enterprise pricing
Top CTMS Platforms by Country (2026)
United States
| Platform | Best For | Pricing (Est.) | Resource Link |
| Veeva Vault CTMS | Enterprise pharma | $50K–$200K/year | https://www.veeva.com |
| Medidata CTMS | Data-driven trials | Custom pricing | https://www.medidata.com |
| Oracle Siebel CTMS | Large-scale trials | $100K+/year | https://www.oracle.com |
| Clinical Conductor CTMS | Research sites | $2K–$10K/year | https://www.adherehealth.com |
| RealTime CTMS | Small to mid sites | $1K–$5K/year | https://www.realtime-ctms.com |
United Kingdom
| Platform | Best For | Pricing (Est.) | Resource Link |
| MACRO CTMS | Academic trials | Custom | https://www.infermed.com |
| EDGE CTMS | NHS research | £5K–£25K/year | https://www.edge.nhs.uk |
| OpenClinica | Open-source trials | Free–£10K/year | https://www.openclinica.com |
| Medrio CTMS | Mid-size CROs | £15K+/year | https://medrio.com |
| Castor EDC + CTMS | Hybrid solution | £10K–£40K/year | https://www.castoredc.com |
India
| Platform | Best For | Pricing (Est.) | Resource Link |
| Jeeva Clinical Trials | Local CROs | ₹3L–₹15L/year | https://www.jeevaclinicaltrials.com |
| Clinion CTMS | End-to-end trials | ₹5L–₹20L/year | https://www.clinion.com |
| Bio-Optronics CTMS | Global trials | Custom | https://www.bio-optronics.com |
| OpenClinica | Budget teams | Free–₹5L/year | https://www.openclinica.com |
| RealTime CTMS | Small sites | ₹1L–₹4L/year | https://www.realtime-ctms.com |
Canada
| Platform | Best For | Pricing (Est.) | Resource Link |
| Medidata CTMS | Large trials | Custom | https://www.medidata.com |
| Veeva Vault CTMS | Enterprise | $60K+/year | https://www.veeva.com |
| Castor EDC | Hybrid usage | $10K–$30K/year | https://www.castoredc.com |
| Florence eBinders + CTMS | Document-heavy trials | $5K–$20K/year | https://florencehc.com |
| OpenClinica | Budget-friendly | Free–$8K/year | https://www.openclinica.com |
Australia
| Platform | Best For | Pricing (Est.) | Resource Link |
| Medrio CTMS | CROs | $15K–$50K/year | https://medrio.com |
| Clinion CTMS | End-to-end | $10K–$40K/year | https://www.clinion.com |
| Veeva Vault CTMS | Enterprise | $70K+/year | https://www.veeva.com |
| OpenClinica | Small teams | Free–$7K/year | https://www.openclinica.com |
| TrialKit CTMS | Mobile trials | $8K–$25K/year | https://www.crucialdatasolutions.com |
Pros and Cons of CTMS
Advantages
- Improved efficiency and automation
- Better compliance and audit readiness
- Real-time tracking and reporting
- Enhanced collaboration
Disadvantages
- High initial cost
- Implementation complexity
- Training required for staff
Common Mistakes When Choosing CTMS
- Ignoring integration capabilities
- Choosing based only on price
- Not considering scalability
- Overlooking user experience
- Skipping vendor support evaluation
Future Trends in CTMS (2026 & Beyond)
- AI-driven patient recruitment
- Predictive analytics for trial outcomes
- Decentralized clinical trials (DCT)
- Blockchain for data security
- Integration with wearable health tech
FAQ
What does CTMS stand for?
CTMS stands for Clinical Trial Management System, used to manage clinical trial operations.
Who uses CTMS?
Pharma companies, CROs, hospitals, and research organizations use CTMS.
Is CTMS required for clinical trials?
While not mandatory, it is essential for efficiency, compliance, and scalability.
What is the difference between CTMS and EDC?
CTMS manages operations, while EDC collects clinical trial data.
How much does CTMS cost?
Costs range from free (open-source) to $200K+ per year, depending on features and scale.
Final Conclusion
A CTMS (Clinical Trial Management System) is no longer optional in modern clinical research—it’s a necessity. With increasing complexity, regulatory pressure, and global collaboration, adopting the right CTMS can dramatically improve efficiency, compliance, and trial success rates.
Choosing the best platform depends on your budget, scale, and integration needs, but investing in the right CTMS will ultimately save time, reduce risk, and accelerate innovation in clinical trials.